Tag: Research
-

One Nucleus Awards Gala
Last Thursday 19th of March brought the UK-wide life sciences ecosystem leaders together for a very special occasion of the #ONAwards26 celebration at No.11 Cavendish Square in London. It was a great honour to have the project between Cellestial and The University of Manchester, supported by a Parkinson’s UK grant, recognised as one of the finalist entries in the “Best Industry and…
-

Preparing for Success
On 10th of March at Jesus College Cambridge, Sygnature Discovery held a seminar on 𝘚𝘵𝘰𝘳𝘪𝘦𝘴 𝘰𝘧 𝘋𝘳𝘶𝘨 𝘋𝘪𝘴𝘤𝘰𝘷𝘦𝘳𝘺 𝘚𝘶𝘤𝘤𝘦𝘴𝘴, joined by the Cellestial team Nat Hastings, Lewis Pennicott, and Peter Bach. Examples of rapid development were discussed where lead optimisation to human trial readiness was complete in only 18 months. Programs that utilise time and…
-

All Roads Lead to Rome: Alfasigma Innovation Day
Nat Hastings and Peter Bach of Cellestial Health found themselves at a beautiful Alfasigma CDMO facility in Pomezia last week as part of the Proventa International Innovation Day. When should manufacturing, formulation, and clinical supply be considered in the course of therapeutics development?It became clear that no matter what stage the product is, the answer…
-

Back at it again in Boston
This week, Dr Nat Hastings represented innovative research into astrocyte pathology in Parkinson’s at the 14th Alzheimer’s & Parkinson’s Drug Development Summit on behalf of Cellestial Health at Hyatt Regency Boston Harbor. Astrocytic lineage is one of the most populous cell types in the brain, yet the damage Parkinson’s pathology does to it remains underappreciated.…
-

BMCS and Parkinson’s UK: New Approaches to the Treatment of Parkinson’s
Dr Nat Hastings represented Cellestial Health at the RSC BMCS and Parkinson’s UK “New Approaches to the Treatment of Parkinson’s” conference on Wednesday 12th, alternating between the corporate exhibitor stand, academic poster presenter, and an audience member of the many insightful talks at the majestic Royal Society of Chemistry halls. Here are the highlights of…
-

CRL Science Day: There is No Such Thing as a Model of Parkinson’s
On Wednesday 24th of September Dr Nat Hastings presented at the Charles River Labs Science Day at Chesterford Research Park on a topic that is relevant to Cellestial’s drug development activities and which stirred many discussions – there is no such things as a model of Parkinson’s disease. What does that mean, when many so-called…
-

Parkinson’s UK: Further Funding for Pioneering Approach
Parkinson’s UK awarded Cellestial Health £55,000 to continue exploring networks of brain cells that may hold answers for new treatments for Parkinson’s. There are many different types of cells in the brain. We know that in Parkinson’s, brain cells that produce dopamine become damaged and start to die over time. However, it’s possible that other…
-

Topic in Spotlight: Brain Delivery
A drug has to be efficacious, safe, and potent – but first of all, it has to get to the right place. On Thursday 10th of July, the Formulation Adviser to Cellestial Health, Prof Claire Thompson of Agility Life Sciences, presented on the topic of formulation choice for proof-of-concept studies in Parkinson’s models at the Bionow…
-
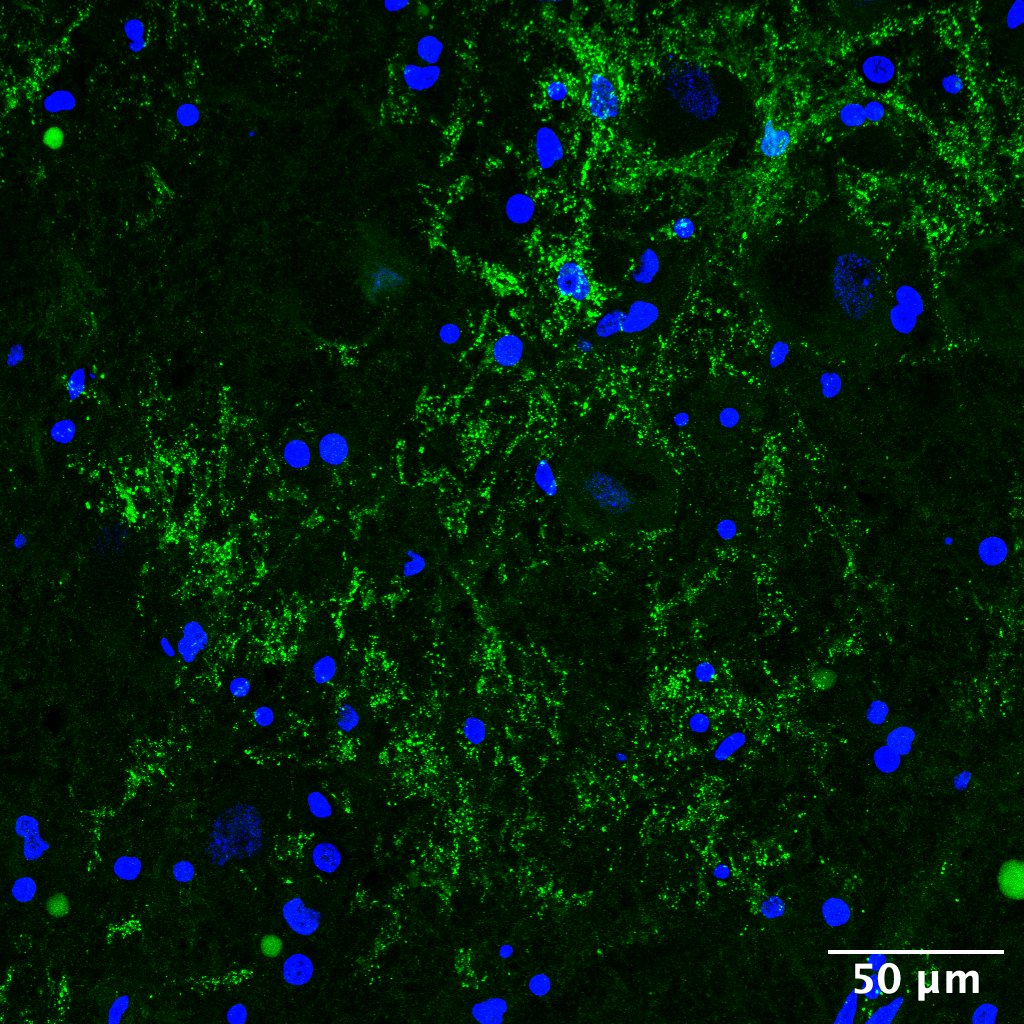
New Paper: Connexin43 is Downregulated in Human Parkinson’s
Translational research often commences in various models – cells, rodents, slice cultures.. But results from all of these rest on an 𝙖𝙨𝙨𝙪𝙢𝙥𝙩𝙞𝙤𝙣 that the underlying cause of the human disease is correctly replicated in the chosen model. There is no single easy way to prove this assumption, especially in a complex and often multi-factorial condition…
-

Making it Happen: a Path to Commercialisation
What does it take to successfully commercialise healthcare innovation? Excellent science is key, but it is not enough. Many factors must converge for a therapeutic discovery to find its way to practical applications: Dr Chloe Hall spoke with Dr Nat Hastings about translatability of Parkinson’s models and specific challenges that emerging companies face in this…
